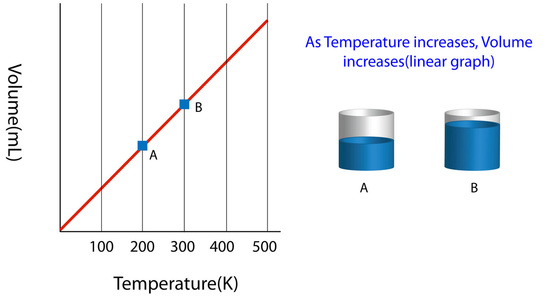
This law was first published by French philosopher Joseph Gay-Lussac in 1802. Charles reenacted the expieriment performed by Joseph Gay Lussac and Tienne Montglofier by using fire to inflate a balloon and watch as it left earths surface. Charles noticed that the volume of gas is directly proportional to its temperature. Lussac credited this discovery to Charles in the 1780's. Charles Law is the relationship between temperature and volume at a constant pressure. As the temperature to heat the gas increases the molecules in the gas start to move around faster and start hitting the container with more force, much like the molecules reacting to high pressure forces in Boyle's law. When the molecules move more quickly and hit the container walls the volume will increase. As the temperature increases the volume increases and the pressure remains constant. If the temperature of a volume of a gas doubles, the volume doubles. Charles law only applys to the behavior of ideal gases not real ones.
Charles Law Formula:
V1/T1=V2/T2
This law was then used later on to determine the volume or temperature of a gas. Equations like the one below are now used.(Temperature must be in complete units i.e K)
At 29.0 degrees a sample of nitrogen gas has a volume of 39.0L. What is the volume of this gas at 50.0 degrees?
T1= 29.0+ 273.15=302.15K
V1=39.0L
T2=50.0+273.15K
V2=?
BY using the formula V1/T1=V2/T2 and transforming it to T2V1/T1=V2 you should end up with the answer V2= 41.8L
Charles law can be found in many day to day things, such as weather balloons. When weather balloons rise to higher altitudes the temperature of the air decreases. The pressure of the balloon decreases and the volume of the gas increases because they are inversely proportional.
Charles Law Formula:
V1/T1=V2/T2
This law was then used later on to determine the volume or temperature of a gas. Equations like the one below are now used.(Temperature must be in complete units i.e K)
At 29.0 degrees a sample of nitrogen gas has a volume of 39.0L. What is the volume of this gas at 50.0 degrees?
T1= 29.0+ 273.15=302.15K
V1=39.0L
T2=50.0+273.15K
V2=?
BY using the formula V1/T1=V2/T2 and transforming it to T2V1/T1=V2 you should end up with the answer V2= 41.8L
Charles law can be found in many day to day things, such as weather balloons. When weather balloons rise to higher altitudes the temperature of the air decreases. The pressure of the balloon decreases and the volume of the gas increases because they are inversely proportional.

