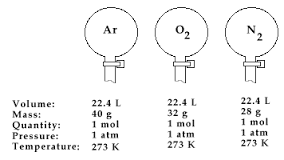
The announced findings of Gay- Lussacs law was only a few years after John Dalton proposed his atomic theory. The link between these two discoveries was recognized by Amadeo Avogadro in 1811. Avogadro came to the conclusion that Gay-Lussacs law could be done by assuming the equivalent volume amounts of different gases under similar conditions all contain the same number of particles. In recognition of Avagadro's contribution to molecular theory 6.0221415 X 10 (to the power of 23) which is the number hat represents number of molecules in one mol) was named Avogadro's number. Avogadro's hypothesis clearly states "equal volumes of all gases, at the same temperature and pressure, have the same number of molecules". This is represented by the formula V=aN. "a" is the constant, "V" is the volume of gas, and "N" is the number of gas molecules. Avogadro's number is 6.0221415 X 10 (to the power of 23). This number is the number of molecules of any gas in a volume of 22.41L. Avogadro hypothesized that two samples of ideal gas which have the same volume, temperature and pressure containing the same number of molecules. Avogadro's hypothsis is important because you can measure the amount of any gas based on knowing the pressure, volume and temperature.
This law was used later on to find the number of molecules in a gas, for example:
Calculate the number of molecules in 0.00273moles NaCl
0.00273moles X 6.0221415 X 10 (to the power of 23)/1mol=1.64 X 10 (to the power of 21) molecules.
Avogadro's Hypothesis can be used in every day life. Such as :
A helium filled balloon weighs much less than a balloon filled with air. Avogadro's Law tells us that equal volumes contain equal numbers of molecules, when pressure and temperature are held constant. Since both balloons contain the same number of molecules, helium atoms still have lower mass than oxygen or nitrogen molecules in air, therefore helium balloon is lighter.
This law was used later on to find the number of molecules in a gas, for example:
Calculate the number of molecules in 0.00273moles NaCl
0.00273moles X 6.0221415 X 10 (to the power of 23)/1mol=1.64 X 10 (to the power of 21) molecules.
Avogadro's Hypothesis can be used in every day life. Such as :
A helium filled balloon weighs much less than a balloon filled with air. Avogadro's Law tells us that equal volumes contain equal numbers of molecules, when pressure and temperature are held constant. Since both balloons contain the same number of molecules, helium atoms still have lower mass than oxygen or nitrogen molecules in air, therefore helium balloon is lighter.
