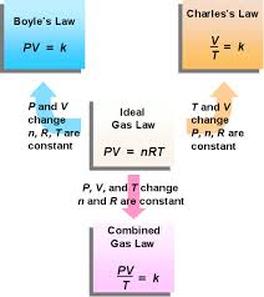
There is no official founder of the combined gas law. The combined gas law is made up of Boyles law (1662), Charles law(1780's) and Gay Lussacs law (early 1800's). The combined gas law is a law used to describe the behavior of a constant amount of gas when pressure, volume and/or temperature is allowed to change. The combined gas law is a combination of Boyle's and Charles law. The combined gas law is used when keeping either temperature or pressure constant becomes difficult. When using combined gas law the temperature must be in kelvins, but pressure and volumes can be in any units.
Combined Gas law Formua:
P1V1/TI=P2V2/T2
The combined gas law was used later on in life to determine the pressure volume or temperature of a gas. For example,
A cylinder contain a gas of volume 30 L, at a pressure of 110 kPa and a temperature of 420 K. Find the temperature of the gas which has a volume 40 L at a pressure of 120 kPa.
Vi = 30 L,
Pi = 110 kPa,
Ti = 420 K, Vf = 40 L,
Pf = 120 kPa
Step 1:Substitute the values in the below final temperature equation: Final Temperature(Tf) = PfVfTi / PiVi = (120 x 40 x 420) / (110 x 30) = 2016000 / 3300 Final Temperature(Tf) = 610.91 K
The combined gas law is found in many every day life things. For example, Scuba diving. As he diver dives deeper into the water the pressure of the gas inside the tank increases. Temperature decreases as the diver goes deeper. The pressure of the scuba tank increases the volume of the gas decreases.
Combined Gas law Formua:
P1V1/TI=P2V2/T2
The combined gas law was used later on in life to determine the pressure volume or temperature of a gas. For example,
A cylinder contain a gas of volume 30 L, at a pressure of 110 kPa and a temperature of 420 K. Find the temperature of the gas which has a volume 40 L at a pressure of 120 kPa.
Vi = 30 L,
Pi = 110 kPa,
Ti = 420 K, Vf = 40 L,
Pf = 120 kPa
Step 1:Substitute the values in the below final temperature equation: Final Temperature(Tf) = PfVfTi / PiVi = (120 x 40 x 420) / (110 x 30) = 2016000 / 3300 Final Temperature(Tf) = 610.91 K
The combined gas law is found in many every day life things. For example, Scuba diving. As he diver dives deeper into the water the pressure of the gas inside the tank increases. Temperature decreases as the diver goes deeper. The pressure of the scuba tank increases the volume of the gas decreases.

